Introduction
Embryo thawing is the reverse of the freezing process, and involves warming the embryos to room temperature to allow the transfer back into culture media at 37°C in CO2 incubator. The embryos are then ready for transfer to the uterus. The embryos are thawed either the day before or on the day of the scheduled embryo transfer.
Embryo thawing takes approximately 2 hours. Sometimes individual cells within the embryo are damaged by the freezing process. Embryos with some freeze damage can still go on to produce a healthy pregnancy however the more the embryo is damaged the less likely it will be for the embryo to develop. Sometimes all of the cells within the embryo are damaged. In this situation the embryo will not be transferred.
Purpose
- Once a couple has gone through an embryo transfer, any extra embryos that were not transferred and have made it to the cleavage or blastocyst stage (day 5) are frozen by a process called vitrification.
- Extending the possibility for pregnancy when fresh cycles fail or when couples want additional children after a successful embryo transfer.
- Offering an alternative to couples that might transfer too many embryos and risk a multiple gestation pregnancy.
- Avoiding embryo wastage by freezing embryos individually for efficient use.
- Increasing pregnancies per retrieval cycle with normal outcomes by 10-30% more. Many studies have evaluated the children born from frozen embryos. The result has uniformly been positive with no increase in birth defects or development abnormalities.
- It enables a precautious policy for embryo transfer.
- This also establishes a cumulative pregnancy rate, increasing the overall chances for patients to conceive per IVF cycle.
- Helping patient management with complications such as Ovarian Hyperstimulation Syndrome (OHSS) and thus to eliminate or decrease the risk of ovarian hyperstimulation syndrome.
- It is less expensive and less invasive than repeating an ovarian stimulation required for a fresh embryo transfer (ET).
- To enhance pregnancy yields from a single ovarian stimulation cycle.
- There is no risk of ovarian hyperstimulation.
Embryo Survival Rate and Quality
Thaw survival rate was calculated as the number or viable or developing embryos on the day of transfer divided by the total number thawed. Some researcher defined embryo survival based on the number of viable cells in an embryo after thawing. An embryo has “survived” if >50% of the cells are viable. We consider an embryo to “partially survive” if <50% of its cells are viable, and to be “atretic” if all the cells are dead at thaw.
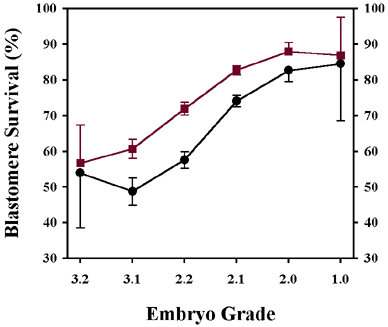
Embryo morphology (appearance of the cells / percentage of fragmentation) is one of the most influential factors for embryo survival. Embryos that are 2, 4, or 8 cells when frozen have about 5-10% greater survival than embryos with an odd number of cells.
Some studied reported that survival rate was 85% for D3 embryos, and 88% for blastocysts (Mary E.P. et al., 2011).Veek et al. (2004) reported that 76.3% of blastocysts survived thawing. Langley et al. (2001) compared thawed day 3 embryos and blastocysts and reported a higher survival rate and subsequent implantation rate for blastocysts.
Pregnancy and Live Birth Rate
Implantation rate was defined as the number of gestational sacs per number of embryos transferred. Live birth was defined as a fetus exiting the maternal body and showing signs of life. In current state of the ART, early embryos having undergone cryopreservation implant at the same rate as equivalent fresh counterparts (EdgarD. H. & Gook D. A.,2012).The outcome from using cryopreserved embryos has uniformly been positive with no increase in birth defects or development abnormalities (Kopeika, J.et al., 2014). In fact, pregnancy rates are increased following frozen embryo transfer, and perinatal outcomes are less affected, compared to embryo transfer in the same cycle as ovarian hyperstimulation was performed (Schnorr J.A. et al., 2001).
The endometrium is believed to not be optimally prepared for implantation following ovarian hyperstimulation, and therefore frozen embryo transfer avails for a separate cycle to focus on optimizing the chances of successful implantation (Schnorr J.A. et al., 2001).
Furthermore, pregnancy rates are similarly affected by complex relationships and like embryo survival only 7-10% of the predictive value can be observed and measured. Age is not a significant factor with frozen embryos but fewer older women have frozen embryos. From the approximately 20 factors reviewed, the most important factors predicting pregnancy rates are the number of surviving embryos transferred, the number of 100% surviving embryos transferred, and the morphology scores of the transferred embryos. The delivered pregnancy rates ranged from 5% (a single poor quality embryo) to 36% (for high quality embryos).
The implantation rate per number thawed was12% for D3 embryos, and 14% for blastocysts. The implantation rate per number of embryos that survived the thaw was 14% for D3 embryos, and 15% for blastocysts. The implantation rate per transfer cycle was 44.6% for D3 embryos, and 38.0% for blastocysts.
Mary E.P. et al ( 2015) reported that for D3 embryos, there were a total of 176 singletons, 53 sets of twins and 2 sets of triplets totalling 288 live births and a 10.0% live birth/thawed embryo rate. For blastocysts, there were 37 singletons, 11 sets of twins and 0 triplets, yielding a total of 59 live births and an 11.7% live born/thawed embryo rate.
The Genetics & IVF Institute is located in the Washington reported that blastocysts (embryos cultured for 5 days rather than 2-3) are a special case. The embryos are much larger and have special needs with regard to freezing without damage. Many centres have had trouble with blastocyst cryo-survival and pregnancy rates. A new protocol developed such as Vitrification with closed system led to a transfer rate of 62% and a 35% pregnancy rate per transfer. This important change now makes blastocyst transfer more appealing since excess blastocysts can be expected to yield pregnancy rates comparable to embryos frozen two to three days after retrieved.
References
- Edgar, D. H.; Gook, D. A. (2012). “A critical appraisal of cryopreservation (slow cooling versus vitrification) of human oocytes and embryos”. Human Reproduction Update 18 (5): 536.
- Kopeika, J.; Thornhill, A.; Khalaf, Y. (2014). “The effect of cryopreservation on the genome of gametes and embryos: principles of cryobiology and critical appraisal of the evidence”. Human Reproduction Update 21 (2): 209–227.
- Veeck LL, Bodine R, Clarke RN, Berrios R, Libraro J, Moschini RM, et al. High pregnancy rates can be achieved after freezing and thawing human blastocysts. FertilSteril. 2004; 82:1418–27.
- Langley MT, Marek DM, Gardner DK, Doody KM, Doody KJ. Extended embryo culture in human assisted reproduction treatments. Hum Reprod. 2001; 16:902–8.
- 7 “Genetics & IVF Institute”. Givf.com. Archived from the original on 2009-07-27. Retrieved 2009-07-27.
- Evans, J.; Hannan, N. J.; Edgell, T. A.; Vollenhoven, B. J.; Lutjen, P. J.; Osianlis, T.; Salamonsen, L. A.; Rombauts, L. J. F. (2014). “Fresh versus frozen embryo transfer: backing clinical decisions with scientific and clinical evidence”. Human Reproduction Update 20 (6): 808–821.
- Wennerholm, U. B.; Soderstrom-Anttila, V.; Bergh, C.; Aittomaki, K.; Hazekamp, J.; Nygren, K. -G.; Selbing, A.; Loft, A. (2009). “Children born after cryopreservation of embryos or oocytes: A systematic review of outcome data”.Human Reproduction 24 (9): 2158–2172.
- Mary Ellen Pavone, Joy Innes, Jennifer Hirshfeld-Cytron, Ralph Kazer, and John Zhang. Comparing thaw survival, implantation and live birth rates from cryopreserved zygotes, embryos and blastocysts. J Hum Reprod Sci. 2011 Jan-Apr; 4(1): 23–28. 356
| Last Reviewed | : | 14 April 2016 |
| Writer | : | Sardiana binti Sarmidi |
| Accreditor | : | Krishnan Kanniah |