Introduction
The development of embryo after the fertilisation of single oocyte and sperm, were evaluated through their morphology. The evaluation of the embryo is actually an evaluation of embryo quality. The evaluation was performed under light microscope at 200 – 400 x magnification at fixed time intervals. Through this evaluation of the embryo, embryologist will assign a grade to each embryo at their various stages of development and determine which are the most likely to result in a healthy pregnancy.
Embryo grading methods were stage dependent involving those characteristic at each stage of embryo development. Different IVF labs, have their own embryo grading system that might differ with one another.
Normal Embryo vs Abnormal Embryo
1. Normal Embryo Development Stages
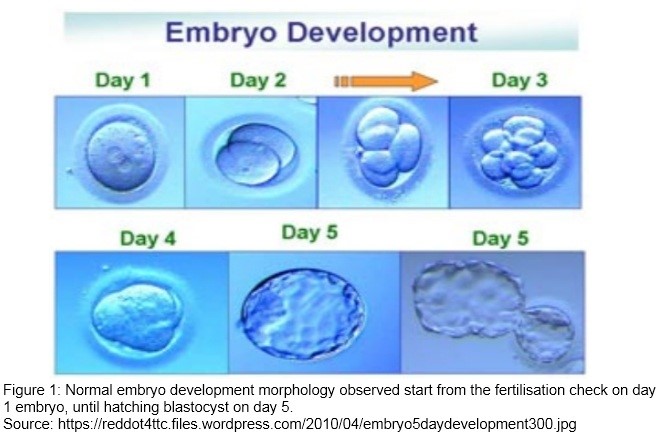
There are several stages of development of embryo after fertilisation before they can be transferred to the uterus. Those stages are marked by cleavage or cell division. Started from a single cell gametes, then divide into many cells as follow:
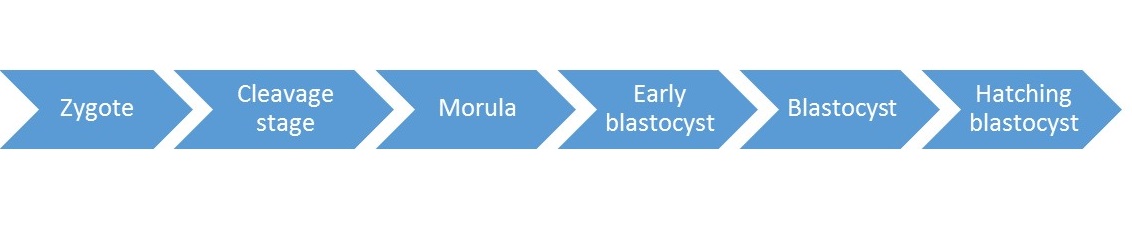 |
| Zygote | : Single cell gamete which are the sperm and the oocyte. Where the fertilisation happen with the presence of 2 pronuclei. |
| Cleavage stage |
: 24hours post fertilisation, it can be 2-4 blastomere (cells formed bycleavage). |
| Morula |
: 96 hours after fertilisation. Compaction process occur where blastomerecontinue dividing and begin to adhere tightly to each other. |
| Early blastocyst |
: The presence of small fluid cavity called blastocoel inside the embryo. |
| Blastocyst |
: Separation between placenta and fetal tissue which normally reach theuterus at the end of fifth day. |
| Hatching blastocyst |
: Embryo will hatch from its zona pellucida, and allows the embryo to implant in the uterus. |
 |
2. Abnormal Embryo Development
Timing Parameters
Developmental timing such as the presence of nuclei, and blastomere at certain timepoints of the embryo is the most important parameters. Which indicate the normal form of embryo represent any chromosome failure or degeneration. The characteristic of embryo observed and classically involved in their evaluation are:
- Cleavage rate
- Blastomere symmetry
- Cytoplasmic appearance
- Extent of Fragmentation
- Blastomere nuclear status
- Trophectoderm (outer layer of blastocyst) quality
- Inner cell mass (ICM) quality
Type of Abnormalities
Abnormalities could be detected started from the observation of their cumulus complex and the shape of the oocyte prior to stimulation and egg collection that has been explained in oocyte abnormalities. In later stage of IVF, the embryo development were observed and classically involved in their evaluation are; cleavage rate, blastomere symmetry, cytoplasmic appearance, extent of fragmentation and blastomere nuclear status.
Pro Nucleus Abnormality
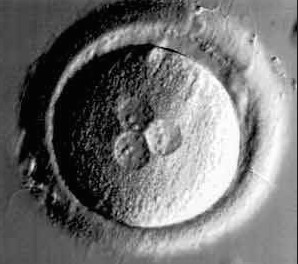 |
Figure 2: 3PN-Presence of 3 Pro Nycleus (PN) observed on fertilisation checked whereas there should be just 2 pro nucleus.
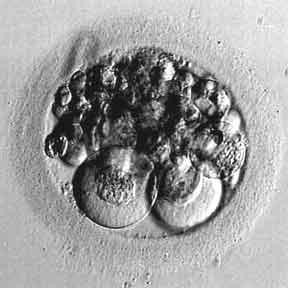
Figure 3: >25% fragmentation that hardly differentiate the presence of the blastomere in the oocyte
Source : http://www.advancedfertility.com/abnorm8c.htm
Fragmentation: portions of the embryo’s cells have broken off through their cleavage process. Preferable to have little or no fragmentation. The more fragmentation seen, the poorer the embryo quality.
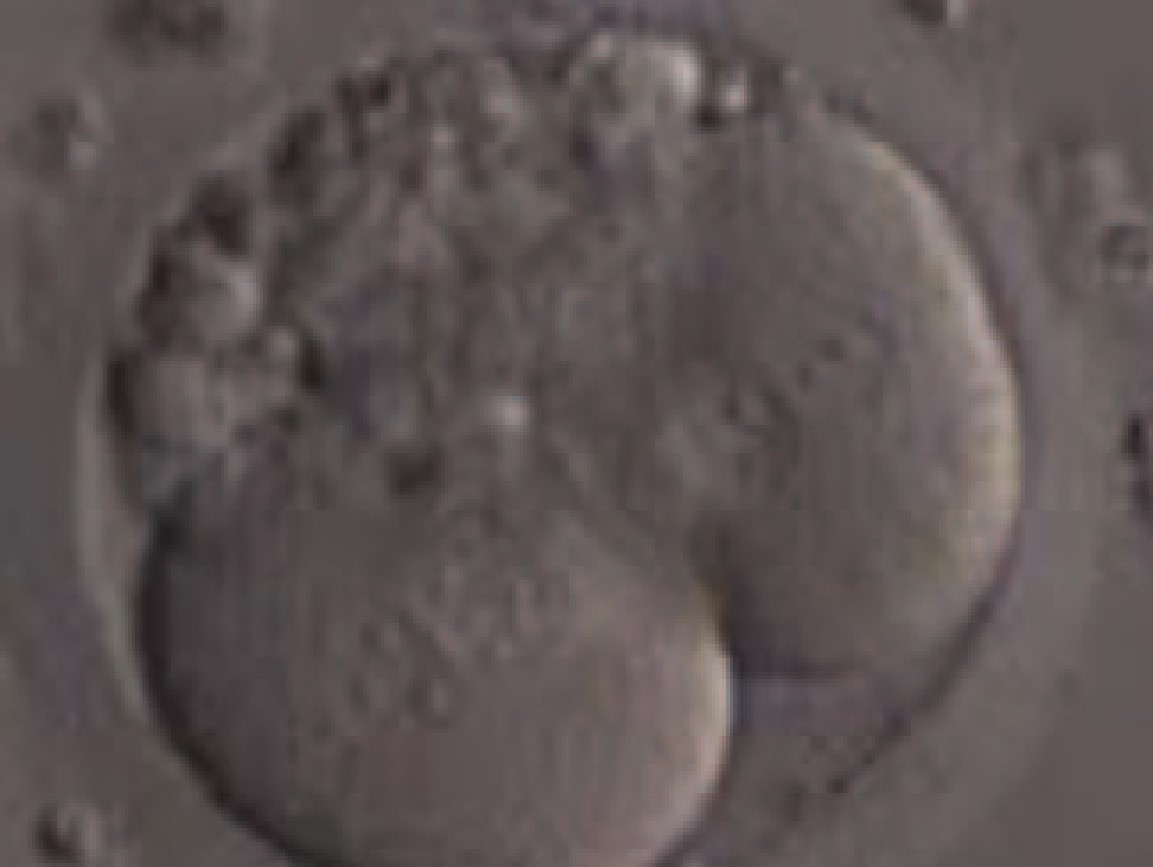 |
Figure 4 : A 2-cell stage embryo with two binucleated and 10 to 20% fragmentation
Multinucleation: Blastomere presented with more than single interphase nucleus and considered abnormal.
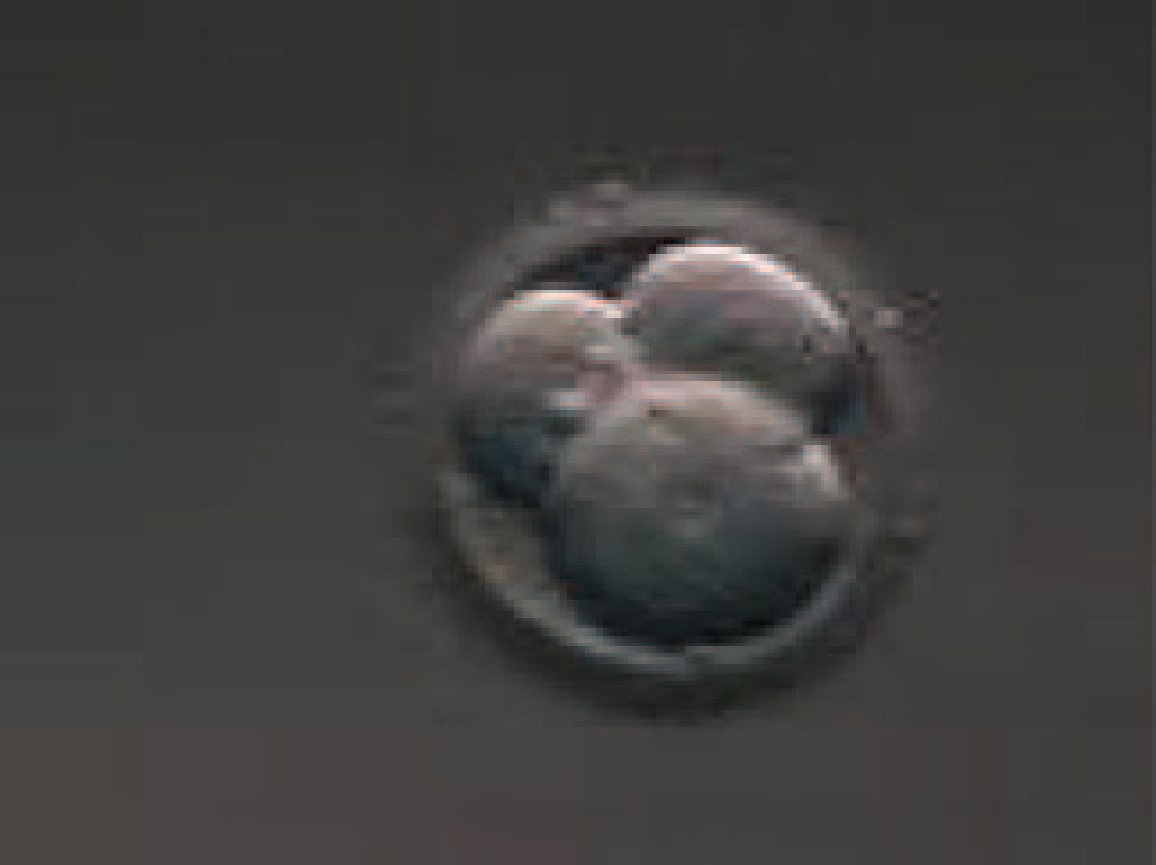 |
Figure 5. A 3 cell embryo with one larger blastomere and two smaller blastomere
Source : Prados et. al., 2012
Cleavage: uneven cell number (3 or 5 cell) and too slow or too fast embryo cleavage (more than 6 cells) on day 2 post insemination
Morphology and Chromosome Abnormalities
Cleavage Stage:
Morphological patterns through day 1 to 3 embryo are important in selecting high potential embryo. Embryo that have not cleaved during 24hours period is called ‘arrested’ embryo. Less than 6 cells on day 3 post insemination are ‘slow’ embryo.
Blastocyst stage:
Higher rates of chromosome abnormalities found in blastocysts developing from embryos that had poor day 3 morphology.
Giant embryo:
Developed from giant oocyte (>200uM in diameter) have been found to be triploid mosaics (Balakier et al., 2002)
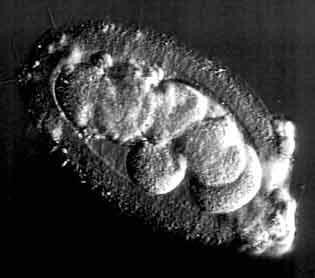 |
Figure 6. An elongated embryo with 7 cells blastomere
Source : http://www.advancedfertility.com/abnorm8c.htm
Elongated embryo:
Similar rates of chromosomal abnormalities as spherical embryos (Magli et al., 2001)
Implication of Abnormal Embryo
Fragmentation, multinucleation, and uneven cell numbers (dysmorphisms) are associated with an increase in abnormalities and decrease implantation potential. Embryos developing slowly or arrested development have higher incidence of post meiotic abnormalities such as mosaicism, monospermic polyploidy and haploidy than normally developing embryo.
Fragmentation:
Fragmentation that less than 10% in day 3 embryos has found to have no significant effect on implantation rate (Van Roven et al., 2001), and a trend was found between the level of fragmentation and incidence of aneuploidy (Munne, 2006). Inconsistent relation between number of blastomere cells and fragmentation with implantation rate has indicated that fragmentation should not be the only morphological criterion assessed.
Multinucleation:
Multinucleation is correlated with high degree of chromosomal aberration and fragmentation and number of blastomere on day 2 and day 3. This abnormalities are also associated with uneven cell size (Hardarson et al., 2001). Embryo with multinucleated blastomeres has been related with lower implantation, pregnancy and birth rates.
Cleavage:
Lundin et al. (2001) have reported that early cleavage at 25 to 27 hours post intracytoplasmic sperm injection (ICSI) of embryo was an independent predictor of live birth. In other report by Ziebe et al. (1997) have shown that 4 cell embryos on day 2 have significant higher implantation rate compared to two and three cell embryo. The development of those blastomere embryo into normal proportion of blastocyst was significantly higher among embryo with 6-8 cell at day 3 compared to less than 6 cells (Alikani et al., 2000).
In later year, Magli et al. (2007) has reported higher incidence of aneuploidy among embryos that were 3-5 cells on day 3 embryo compared to 6-8 cells. Through chromosomal analysis on more than 500 embryos, demonstrated that a highly significant relationship between maternal age and aneuploidy. This chromosomal abnormalities was significantly higher in arrested, slow-cleaving and accelerated embryos compared with normally developing embryos (Magli et al., 2007).
Regardless the quality of blastocyst, more than 40% of mosaics are still chromosomally abnormal and will not implant or will spontaneously abort. Because aneuploidy is not related to cleavage stage dysmorphism and trisomies can reach blastocyst stage and beyond, morphological analysis is not enough to select against chromosome abnormalities, and thus preimplantation genetic diagnosis should be recommended in patients 35 and older.
References:
- Alikani M, Calderon G, Tomkin G, Garrisi J, Kokot M, Cohen J. 2000. Cleavage anomalies in early human embryos and survival after prolonged culture in-vitro. Hum Reprod; 15:2634–2643.
- Balakier, H., Bouman, D., Sojecki, A., Librach, C., Squire, J.A., 2002. Morphological and cytogenetic analysis of human giant oocytes and giant embryos. Hum. Reprod. 17, 2394–2401.
- Hardarson T, Hanson C, Sjo¨ gren A, Lundin K. 2001. Human embryos with unevenly sized blastomeres have lower pregnancy and implantation rates: indications for aneuploidy and multinucleation. Hum Reprod; 16:313–318.
- Lundin K, Bergh C, Hardarson T. 2001. Early embryo cleavage is a strong indicator of embryo quality in human IVF. Hum Reprod; 16:2652–2657.
- Magli MC, Gianaroli L, Ferraretti AP, Lappi M, Ruberti A, Farfalli V. 2007. Embryo morphology and development are dependent on the chromosomal complement. Fertil Steril; 87:534–540.
- Magli, M.C., Gianaroli, L., Ferraretti, A.P. 2001. Chromosomal abnormalities in embryos. Mol. Cell. Endocrinol. 183, 29–34.
- Munne´ S. 2006. Chromosome abnormalities and their relationship to morphology and development of human embryos. Reprod Biomed Online; 12:234–253.
- Prados F.J, Debrock S, Lemmen J.G, Agerholm. 2012. The cleavage stage embryo. Hum.Reprod; 1-22
- Van Royen E, Mangelschots K, De Neubourg D, Laureys I, Ryckaert G, Gerris J. 2001. Calculating the implantation potential of day 3 embryos in women younger than 38 years of age: a new model. Hum Reprod; 16:326–332.
- Ziebe S, Petersen K, Lindenberg S, Andersen AG, Gabrielsen A, Andersen AN. 1997. Embryo morphology or cleavage stage: how to select the best embryos for transfer after in-vitro fertilization. Hum Reprod; 12:1545–1549.
| Last Reviewed | : | 14 April 2016 |
| Writer | : | Siti Norfaizah binti Wagiman |
| Translator | : | Siti Norfaizah binti Wagiman |
| Accreditor | : | Krishnan A/L Kanniah |







