Beauty and cosmetics are synonymous. Today, there are many beauty products easily available in the marketplace.
These beauty products are sold in many forms, colours, flavours, smell and mode of applications with various claims and intended uses. For example, one can use cream A to get a fairer skin or getting slimmer by drinking Juice B or taking capsule C to get a flawless skin. At the same time, consumers are constantly tempted with exciting and alluring advertisements published both in printed and electronic media.
But, do these products really work? Are they safe? Are they being controlled by any authority agency?
What Is A Cosmetic Product?
Cosmetic products are commonly referred to makeup products such as blushers, eye shadows and lipsticks to make someone look beautiful. For regulatory control purposes , cosmetic products are defined as “any substance or preparation intended to be placed in contact with various external parts of the human body (epidermis, hair system, nails, lips and external genital organs) or with teeth and the mucous membranes of the oral cavity, with a view exclusively or mainly to cleaning them, perfuming them, changing their appearance and/or correcting body odors and/or protecting them or keeping them in good condition.” Hence, cosmetic products are not only limited to lipsticks or compact powders but also personal care products such as soaps, shampoos and oral care products.
 |
Figure 1: Examples of cosmetic products
Consumers and public tend to make assumptions that all beauty products are cosmetics regardless of how the product is being used either by external applications or oral consumption. This is not true because a cosmetic product by definition is only allowed for external use. Therefore, any beauty products to be consumed orally are not classified as cosmetics but need to be classified as other categories of products such as health supplements, natural products or food. For example, a product containing collagen can be sold as a cosmetic product when its instruction of use is to be applied on the skin whereas when the product is instructed to be consumed orally, it is then classified as health supplements or food products.
There are two authority agencies under the Ministry of Health Malaysia that control such products namely National Pharmaceutical Regulatory Agency (NPRA) and Food Safety and Quality Division.
 |
Figure 2: Examples of products to be consumed orally as drink that have used cosmetic claims.
Advertisement of Cosmetic Products.
Consumers are advised not to be easily influenced by cosmetics’ advertisements especially those with excessive claims. Most often, these advertisements are promoting cosmetic products together with other beauty and health products to achieve the optimum effects. Inevitably, one would make the unnecessary purchase. These irresponsible advertisements may mislead and confused the consumers.
Figure 3: Example of advertisement with misleading information.
Control of Cosmetic Products in Malaysia
All cosmetic products marketed in Malaysia must first be notified to the NPRA and only notified products may legally be imported or manufactured for sale in the country. Cosmetic products must only contain permitted substances which are used within its allowable limits and conditions. The correct instruction of use for a cosmetic product must be clearly stated. In addition, any advertisements related to cosmetic products must conform to the stipulated guidelines.
NPRA is actively monitoring the cosmetic products placed in the local market to ensure safety and quality as well as conformance to the current guidelines and regulations.
Public and consumers may check the notification status of a cosmetic product from NPRA’s official website through product search QUEST3+ system or through NPRA Product Status Application which can be downloaded through Google Play Store.
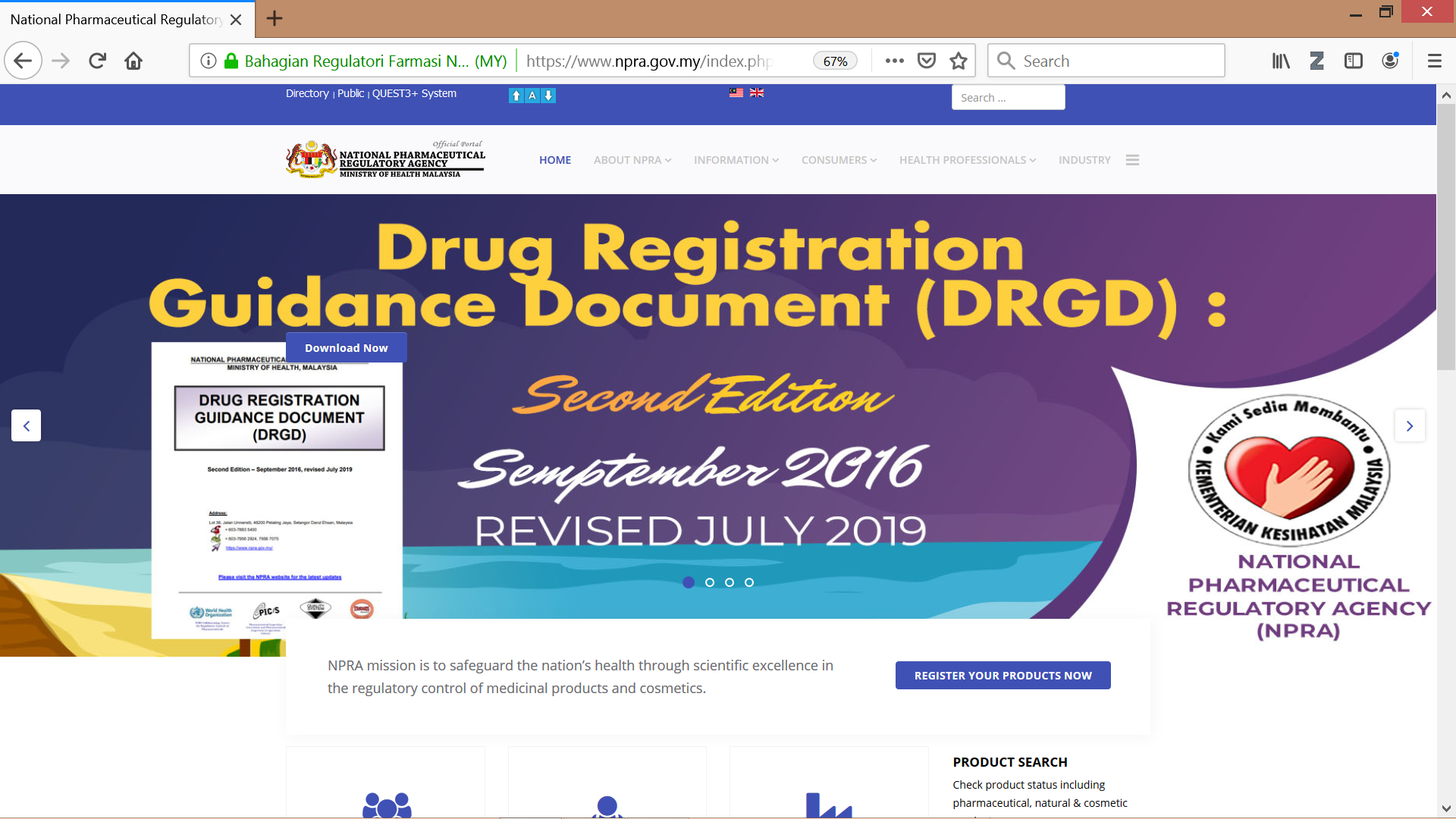
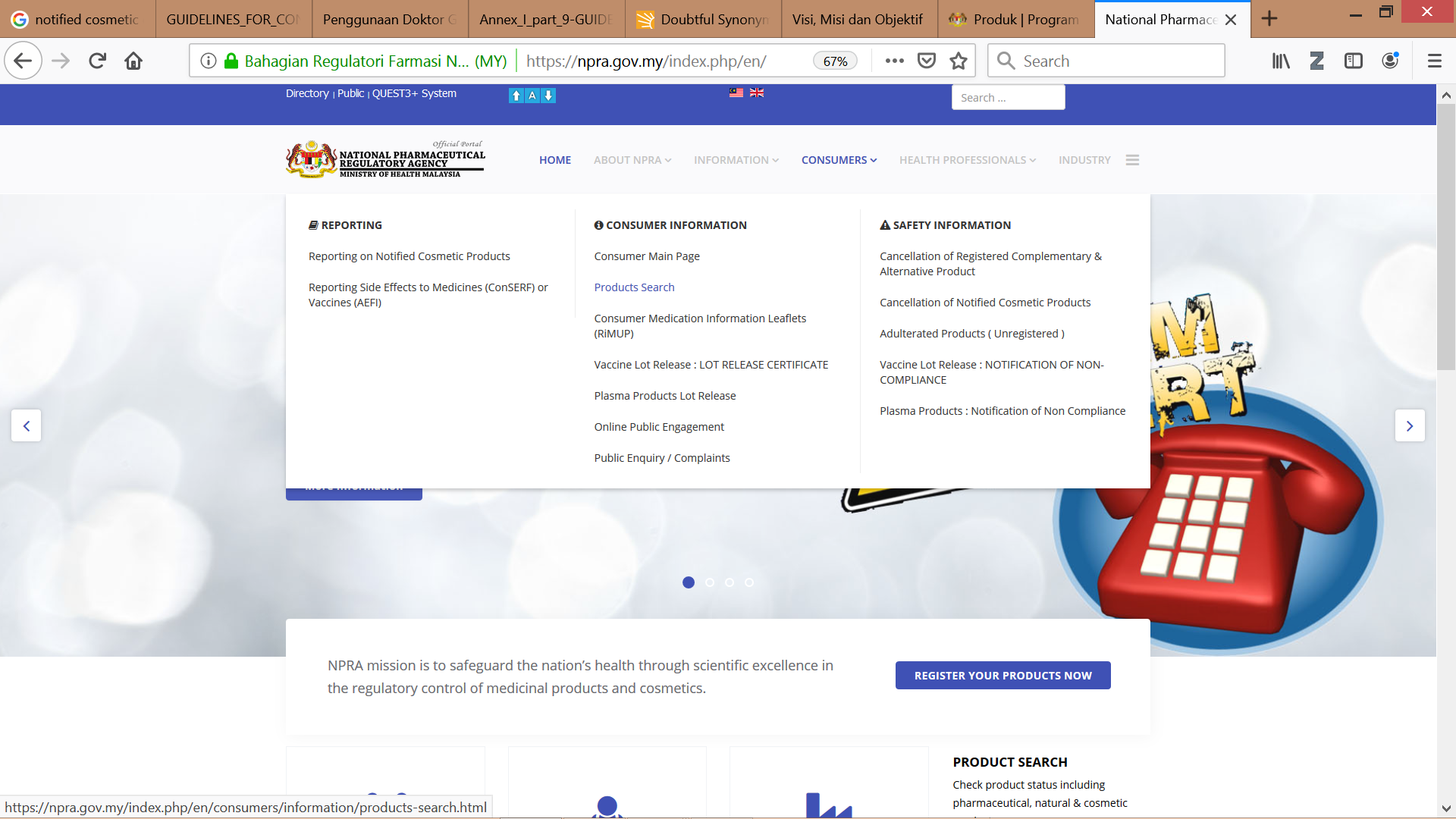
Figure 4 & 5: NPRA’s website www.npra.gov.my
| Last Reveiwed | : | 22 August 2019 |
| Translator | : | Noor Hidayah bt. Mohd Noor |
| Accreditor | : | Zuraida bt. Abdullah |
| Reviewer | : | Wan Mohaina bt. Wan Mohammad |
| : | Hanum Maisarah bt. Abdul Rahman |








